|
3/31/2023 0 Comments Ion bondingthe ions formed are negative, because they have more electrons than protonsįor elements in groups 6 and 7, the charge on the ion is equal to (8 minus group number).The outer shells of non-metal atoms gain electrons when they form ions: A sodium atom loses one electron to form a sodium ion Forming negative ions An atom of sodium will lose an electron and form a positive ion. the ions have the electronic structure of a noble gas (group 0 element), with a full outer shellįor elements in groups 1, 2 and 3, the number of electrons lost is the same as the group number. Ionic bonds are formed between a metal and non-metal, for example sodium chloride.the ions are positive, because they have more protons than electrons.Metal atoms lose electrons from their outer shell when they form ions: non-metal atoms gain electrons to form negatively charged ions.metal atoms lose electrons to form positively charged ions. 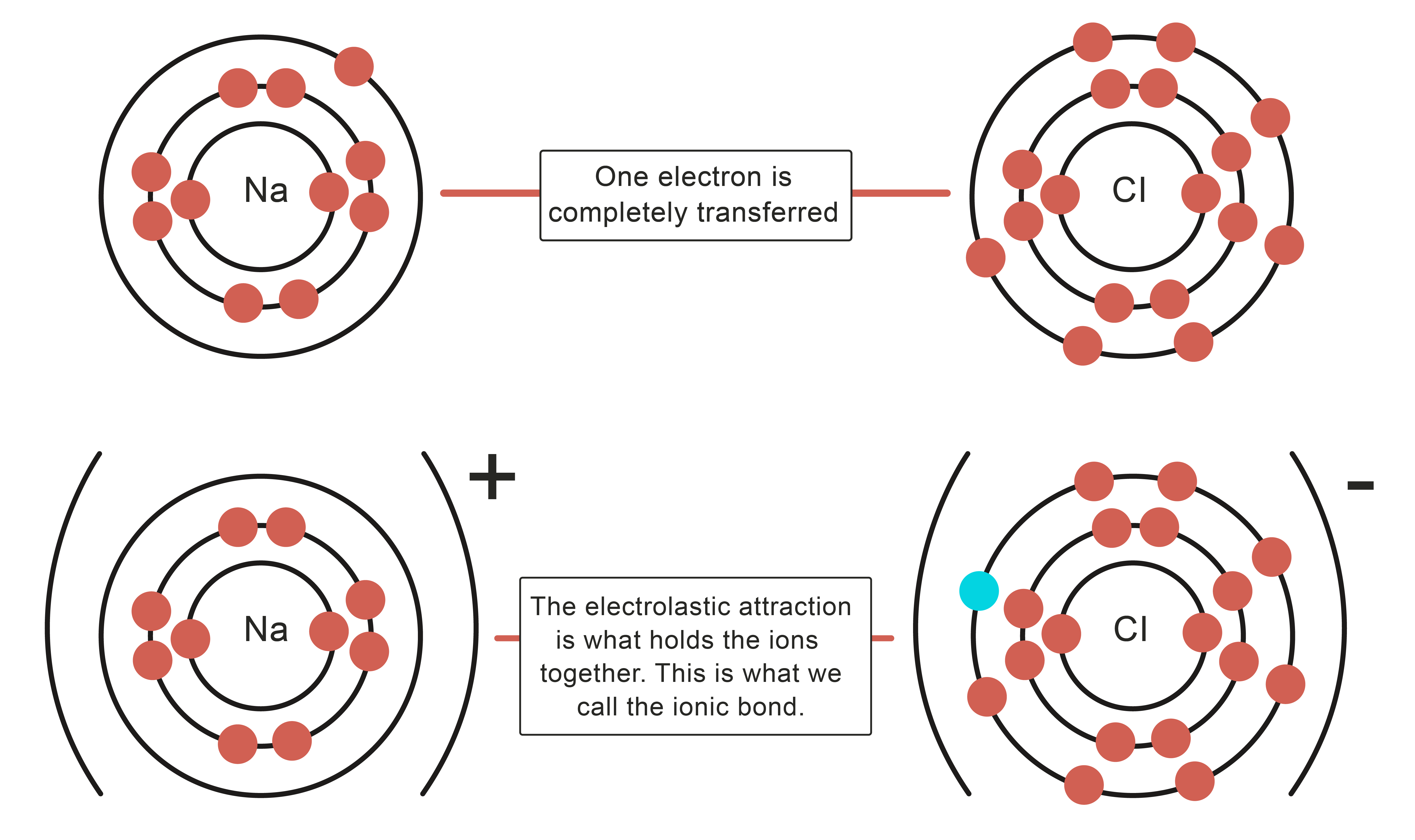
Ions form when atoms lose or gain electrons to obtain a full outer shell: 10.An ion is an atom or group of atoms with a positive or negative charge. 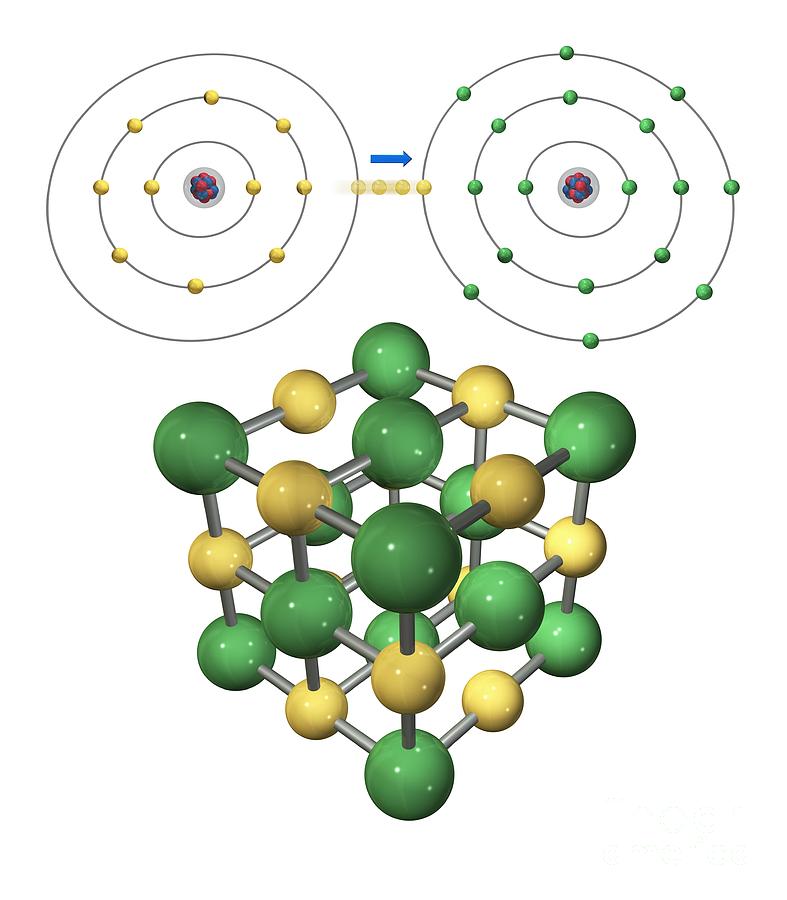
or molecular compounds form when elements share electrons in a covalent bond to form molecules. 10.1016/S0166-1280(03)00029-0 Crossref, Google Scholar Ionic Compounds Containing a Metal and a Polyatomic Ion. 10.1103/PhysRevB.70.235121 Crossref, Google Scholar Ions are atoms that have gained or lost one or more electrons to form a charged particle. 10.1103/PhysRevB.49.16223 Crossref, Google Scholar DEFINITION: The formation of an IONIC BOND is the result of the transfer of one or more electrons from a metal onto a non-metal. Electrons transfer from sodium atoms to adjacent chlorine atoms, since the valence electrons in sodium are loosely bound and chlorine has a large electron affinity. 10.7566/JPSJ.83.124713 Link, Google Scholar A prototypical example is the sodium chloride crystal, as we discussed earlier. Ionic bonding is the attraction between positively- and negatively-charged ions. 10.1103/PhysRevB.24.864 Crossref, Google Scholar The steps involved in the formation of ionic bond can be summarized as: a) An electropositive atom (metal) loses electron(s) to form a positively charged ion. Na + (H2O)n Figure 11.2.1: Ion-Dipole interaction. Note, these must be for solutions (and not pure substances) as they involve two different species (an ion and a polar molecule). 10.1016/j.jpowsour.2011.09.064 Crossref, Google Scholar Ion-Dipole Forces are involved in solutions where an ionic compound is dissolved into a polar solvent, like that of a solution of table salt (NaCl) in water. 10.1016/j.elecom.2012.02.020 Crossref, Google Scholar Ionic bonding is a type of chemical bonding that involves the electrostatic attraction between oppositely charged ions, or between two atoms with sharply. The spectra were obtained at four detachment photon energies: 2.33, 3.49, 4.66, and 6.42 eV. 10.1038/nmat3309 Crossref, Google Scholar Ab-initio methods ABSTRACT The structure and bonding of aluminum oxide clusters, Al x O y ( x 12, y 15), are studied with anion photoelectron spectroscopy (PES) and are compared with preliminary ab initio calculations. Ionic bonding Sodium Chloride, IGCSE & GCSE Chemistry revision notes. Ionic bonding is a kind of chemical bonding that arises from the mutual attraction of oppositely charged ions.  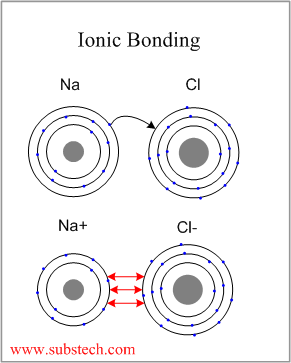
10.1149/2.007305jes Crossref, Google Scholar Ionic Bonds between Group I & Group VII Elements. 10.1038/nmat2920 Crossref, Google Scholar
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
